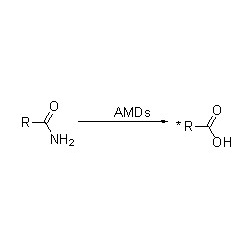
Amidase (AMD)
Li-enzyme:Ke li-catalyst tsa baeloji tse kholo, li-enzyme tse ngata ke liprotheine
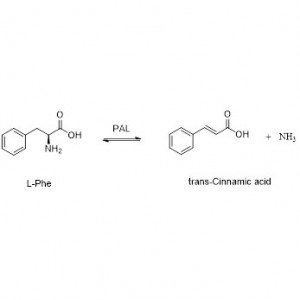
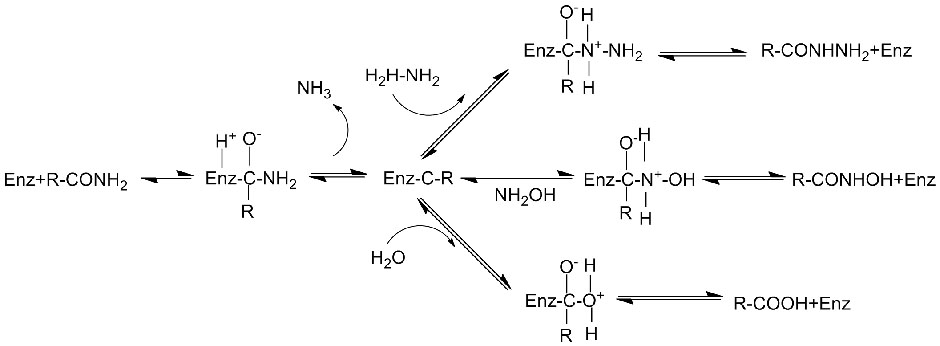
Bohareng ba 'mele:Kenya hydrolysis ea li-amide tse fapaneng tsa tlhaho le tsa kantle ho naha le tse nang le monko o monate ka ho fetisetsa sehlopha sa acyl metsing ka tlhahiso ea li-acid tse lokolohileng le ammonia. Li-acid tsa Hydroxamic le li-acid tse ling tsa organic li sebelisoa haholo e le lithethefatsi hobane ke likarolo tsa lintlha tsa kholo, lithibela-mafu le li-antitumor inhibitors. Li-amidase li ka aroloa ka li-acylase tsa mofuta oa R le mofuta oa S ho latela khetho ea catalyst.
Ntle le ho susumetsa hydrolysis ea amides, amidase e ka boela ea susumetsa liketso tsa phetisetso ea acyl moo ho nang le li-substrate tse ling tse kang hydroxylamine.
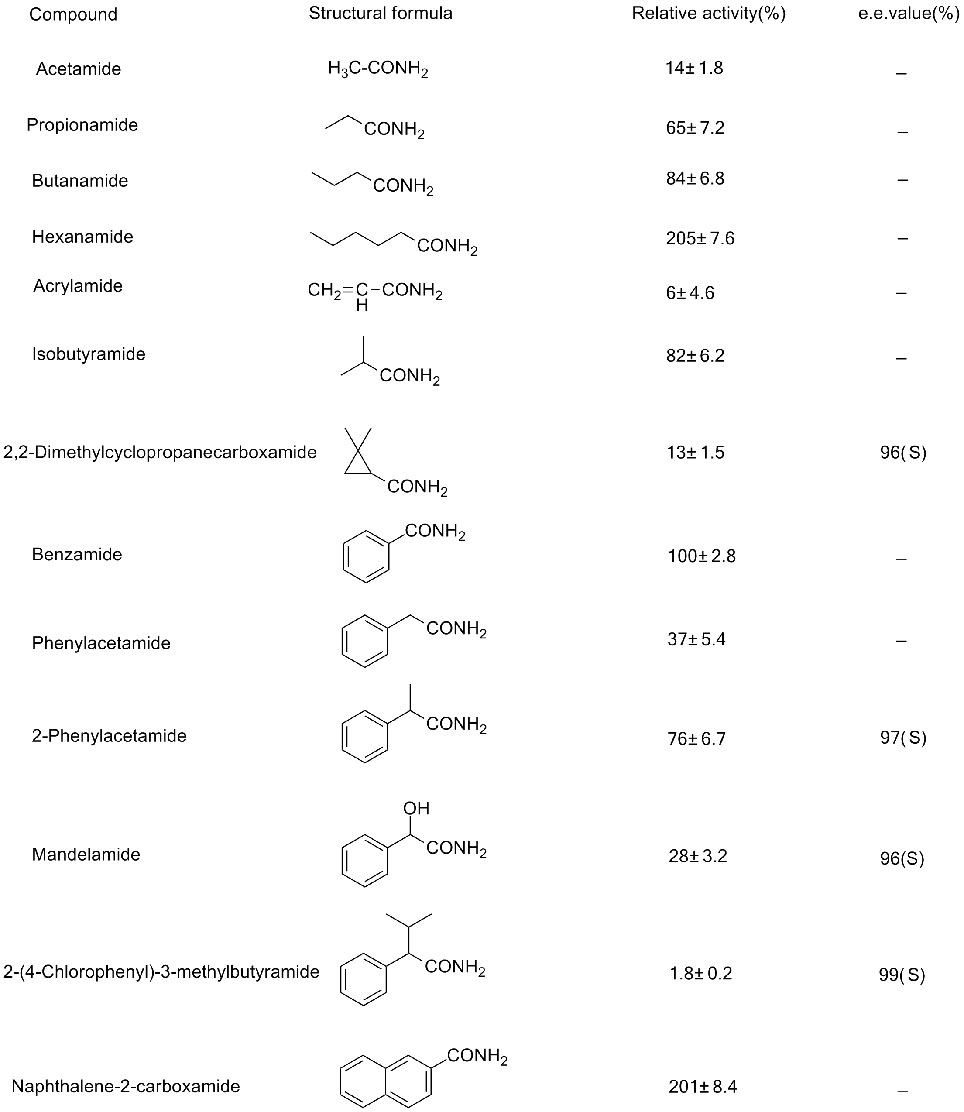
Amidase e nang le mehloli e fapaneng e na le boikgethelo bo fapaneng ba substrate, tse ling tsa tsona li ka hydrolyze amides ea aromatic feela, tse ling tsa tsona li ka hydrolyze aliphatic amides feela, 'me tse ling li ka hydrolyze α-kapa ω-amino amides. Bongata ba li-amine li na le ts'ebetso e ntle ea catalytic feela bakeng sa amides ea acyclic kapa e bonolo ea aromatic, empa bakeng sa li-aromatics tse rarahaneng, li-amides tsa heterocyclic, haholo-holo li-amides tse nang le ortho substituents, hangata li na le ts'ebetso e tlase (ke li-enzyme tse 'maloa feela tse bonts'ang litlamorao tse betere tsa catalytic).
Mokhoa oa ho kenya tšebetsong:
| Li-enzyme | Khoutu ea Sehlahisoa | Khoutu ea Sehlahisoa |
| Phofo ea Enzyme | ES-AMD-101~ ES-AMD-119 | sete ea li-amidase tse 19, 50 mg ka 'ngoe Lintho tse 19 * 50mg / ntho, kapa bongata bo bong |
| Sete ea ho Hlahlobisisa (SynKit) | ES-AMD-1900 | sete ea li-amidase tse 19, 1 mg ka 'ngoe Lintho tse 19 * 1mg / ntho |
★ Ho ikhetha ha substrate haholo.
★ Khetho e matla ea chiral.
★ Bokgoni bo phahameng ba ho fetolela.
★ Lihlahisoa tse fokolang tse setseng.
★ Maemo a bobebe a karabelo.
★ E sireletsa tikoloho.
➢ Tlhahlobo ea li-enzyme e lokela ho etsoa bakeng sa li-substrate tse itseng ka lebaka la ho khetheha ha substrate, 'me ho fumanoe enzyme e susumetsang substrate e reriloeng ka phello e ntle ka ho fetisisa ea ho susumetsa.
➢ Le ka mohla u se ke ua kopana le maemo a feteletseng a kang: mocheso o phahameng, pH e phahameng/e tlase le motsoako oa tlhaho o nang le mahloriso a phahameng.
➢ Ka tlwaelo, sistimi ya karabelo e lokela ho kenyeletsa substrate, tharollo ya buffer (pH e ntle ka ho fetisisa ya karabelo ya enzyme). Di-substrate tse kang hydroxylamine di lokela ho ba teng tsamaisong ya karabelo ya phetiso ya acyl.
➢ AMD e lokela ho eketsoa qetellong tsamaisong ea karabelo e nang le pH le mocheso o motle oa karabelo.
➢ Mefuta eohle ea AMD e na le maemo a fapaneng a karabelo a matle, kahoo e 'ngoe le e 'ngoe ea tsona e lokela ho ithutoa ka bonngoe.
Mohlala oa 1(1):
Mosebetsi oa Hydrolysis ho li-substrate tse fapaneng tsa Amide
| Sebaka se ka tlas'a lefatše | Mosebetsi o itseng μmols motsotso-1mg-1 | Sebaka se ka tlas'a lefatše | Mosebetsi o itseng μmols motsotso-1mg-1 |
| Acetamide | 3.8 | ο-OH benzamide | 1.4 |
| Propionamide | 3.9 | p-OH benzamide | 1.2 |
| Lactamide | 12.8 | ο-NH2benzamide | 1.0 |
| Butyramide | 11.9 | p-NH2benzamide | 0.8 |
| Isobutyramide | 26.2 | ο-Toluamide | 0.3 |
| Pentanamide | 22.0 | p-Toluamide | 8.1 |
| Hexanamide | 6.4 | Nikotinamide | 1.7 |
| Cyclohexanamide | 19.5 | Isonicotinamide | 1.8 |
| Acrylamide | 10.2 | Picolinamide | 2.1 |
| Metacrylamide | 3.5 | 3-Phenylpropionamide | 7.6 |
| Prolinamide | 3.4 | Indol-3-acetamide | 1.9 |
| Benzamide | 6.8 |
Karabelo e entsoe ka tharollo ea 50mM sodium phosphate buffer, pH 7.5, ho 70 ℃.
| Mahareng | Hydroxylamine | Hydrazine |
| Acetamide | 8.4 | 1.4 |
| Propionamide | 18.4 | 3.0 |
| Isobutyramide | 25.0 | 22.7 |
| Benzamide | 9.2 | 6.1 |
Karabelo e entsoe ka tharollo ea 50mM sodium phosphate buffer, pH 7.5, ho 70 ℃.
Khokahano ea reagent e amanang: li-amides, 100 mM(benzamide, 10 mM); hydroxylamine le hydrazine, 400 mM; enzyme 0.9 μM.
Mohlala oa 2(2):
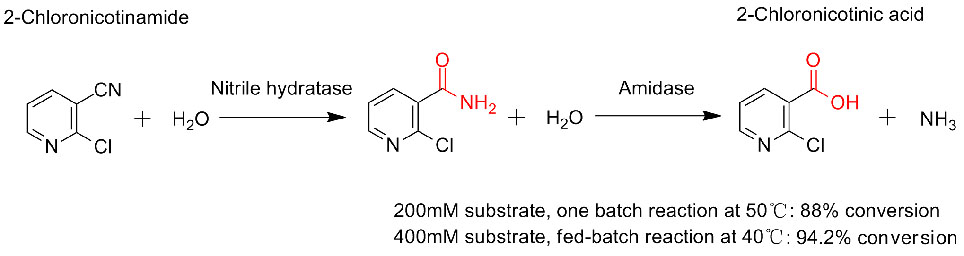
Mohlala oa 3(3):
1. D'Abusco AS, Ammendola S., le ba bang. Extremophiles, 2001, 5:183-192.
2. Guo FM, Wu JP, Yang LR, jj. Process Biochemistry, 2015, 50(8): 1400-1404.
3. Zheng RC, Jin JQ, Wu ZM, jj. Khemistri ea Bioorganic, 2017, E fumaneha inthaneteng 7.